USP (United States Pharmacopoeia) and EP (European Pharmacopoeia) standards, recommend the use of bioindicators during ethylene oxide sterilization processes of pharmaceuticals, drugs in vials, culture media, medical instruments and more.
What is ethylene oxide sterilization?
Ethylene oxide (EtO) sterilization is a low‑temperature gas sterilization method widely used for heat and moisture‑sensitive medical devices that cannot withstand steam or other high‑temperature processes. EtO effectively penetrates complex geometries and packaging, achieving microbial inactivation without damaging delicate materials.
- Self-contained
Self-contained biological indicators are made of a thermoplastic tube, with a cup permeable to ethylene oxide, that contains an ampoule with a growth medium with a pH indicator and a disc impregnated with Bacillus atrophaeus (ATCC 9372) spores in predefined concentrations. They are manufactured under strictly controlled conditions to comply with the current editions of USP and EP, and in accordance with ISO 11138 and EN 866 standards.
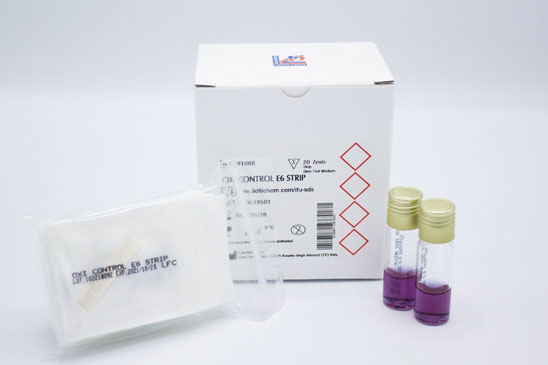
- Strips
These BIs are special filter paper strips, inoculated with Bacillus atrophaeus (ATCC 9372) spores in predefined concentrations and contained in a special envelope. They are manufactured under strictly controlled conditions to comply with the current editions of USP and EP, and in accordance with ISO 11138 and EN 866 standards.
- Spore suspension
Spore suspensions consist of Bacillus atrophaeus (ATCC 9372) spores suspended in distilled water in predefined concentrations. They are manufactured under strictly controlled conditions to comply with the current edition of USP and in accordance with ISO 11138 and EN 866 standards.
View the complete range of Biological indicators Ethylene Oxide
View the complete range of Biological indicators Ethylene Oxide / Dry Heat

