Biological indicators are standardized systems specifically designed to evaluate the effectiveness of
sterilization processes. They consist of a defined population of highly resistant bacterial spores
applied to a carrier which is exposed to the sterilization cycle as a direct microbiological challenge.
By confirming the inactivation of these resistant microorganisms, biological indicators provide
reliable evidence of sterilization performance and are widely used for the validation, routine
monitoring, and periodic verification of sterilization processes in medical, pharmaceutical, and
aseptic applications.
The regulations USP (United States Pharmacopoeia), EP (European Pharmacopoeia) and DAB (Deutsches Arzneibuch) recommend the use of bioindicators during the sterilization processes of drugs, medicines in vials, culture media, medical instruments and others.
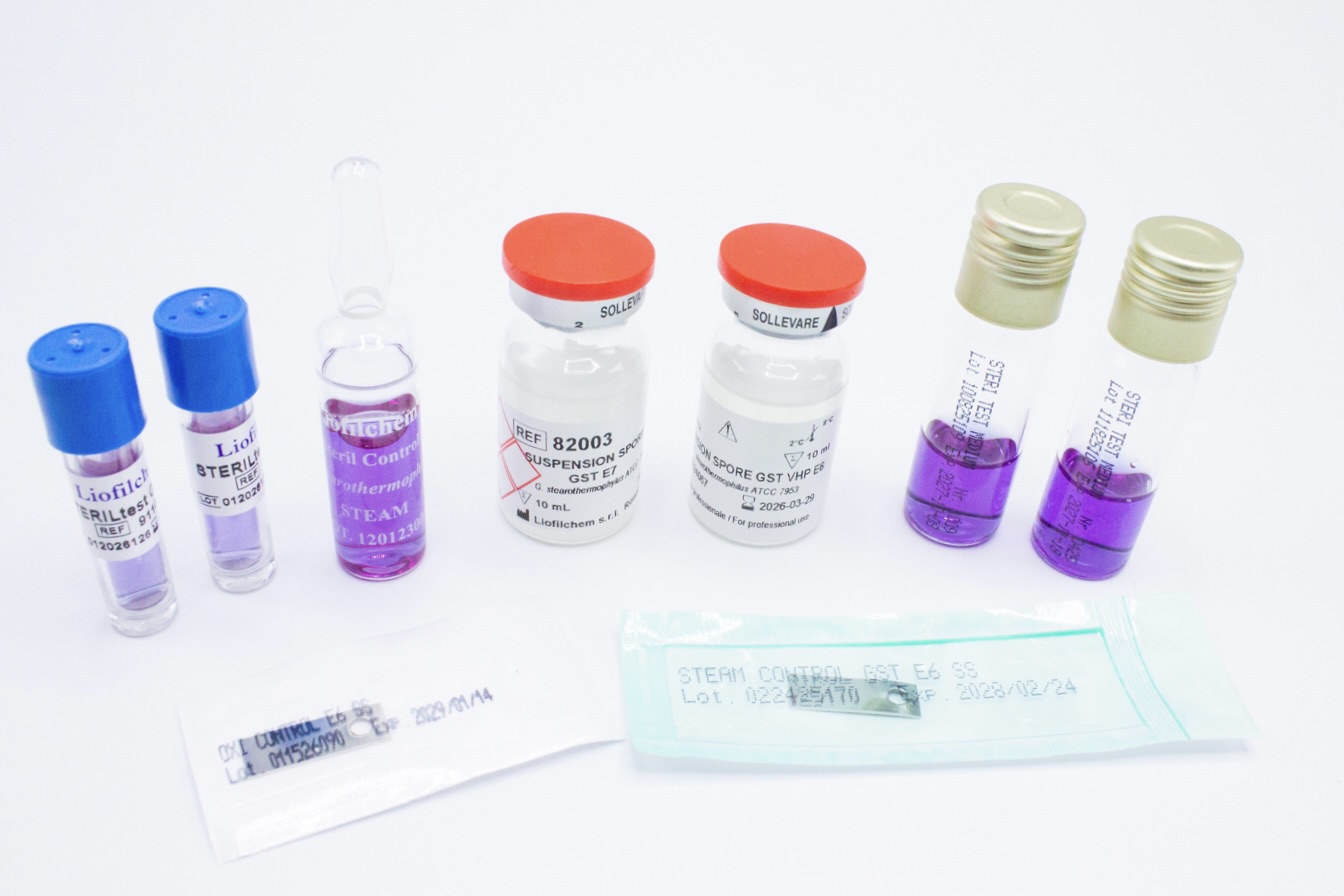
Our Biological Indicators
Biological Indicators manufactured by Liofilchem® are available in ampoule, self-contained, strip,
suspension, and coupon formats, and in a variety of materials including glass, aluminum, Poliflex,
steel, and PVC. Custom-sized Biological Indicators can also be produced upon request.
All Biological Indicators are supplied with a certificate reporting D and Z values, Fbio (Steril
Control GST E6, Steam Control GST E6 SS), survival time, kill time, population, and purity.
Manufacturing is carried out in a strictly controlled environment to ensure compliance with the
requirements of USP and EP, as well as with ISO 11138 and EN 866 standards.